
last updated
Use of a separatory funnel or "How to do an upscale extraction"
A separatory funnel is a standard piece of equipment in synthetic chemistry.

It generally consists of a conical or pear-shaped glass body with a stopcock and a stopper on top. The
conic shape allows for the clean separation of the two layer because of a small interface on the low end. Although it
is easy to use, there are some important points that have to be considered:
1. Stopper
The stopper on top can be made from glass (used above) or Teflon. It is imperative that it fits tightly, so that the solution does not leak out when the separatory funnel is inverted. If a ground glass joint does not fit perfectly, a minute amount of grease is applied to the upper part of the joint to get a better seal.
The stopper has to be removed when draining the lower layer. If the stopper were not removed, a vacuum will build up above the liquid
upon draining. This vacuum will reduce the rate of draining and
ultimately stop it completely. After some time, the vacuum will suck air in (from the stem) and the phases will mix again.
2. Stopcock plug
The stopcock plugs can be made from glass (shown on the left below) or Teflon. It is important to have a good seal here as well. Again, a very small amount of grease can be applied to the glass plugs (shown on the left) to improve the seal and allow for better movement of the plug. The grease should be used sparingly, because it will clog up the hole in the plug!
The glass plug has to be held in place with a metal clip. Teflon plugs usually possess a thread (some glass manufacturers offer those version for glass plugs as well), which allows to place a nut on it to hold it in place. Teflon plugs should not be lubricated!
In order to find out which plug is needed, the glass joint has to be examined. Glass stopcocks require a ground glass joint (looks milky if clean and is rough on the inside). Teflon stopcocks use a polished joint (clear).
Do not attempt to fit a glass stopcock into a
Teflon joint! The slopes on the plugs are slightly different and the joint will break!
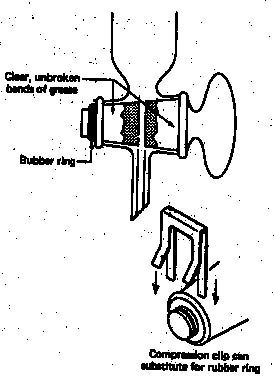

The hole of the plug has to be open and match up with the holes in the stem and separatory funnel.
3. Before you start
Perform the following tests before you start. Suspend the separatory funnel in an iron ring (make sure it does not fall through!).While the stopcock is closed, pour ~20 mL of
a lquid (i.e., water) into the separatory funnel (with a short stem
funnel). Check if the solvent leaks out at the stopcock. If this does
not occur, place the stopper on top and invert the funnel. Does the
lquid leak out now? If not, place the funnel back in the ring stand and remove the stopper. Open the stopcock and drain the solvent. If the solvent does not drain, you probably used too much grease to lubricate the stopcock. Pour the solvent out and remove the stopcock plug and check the hole. If it is clogged, use a wooden applicator or piece of wire to open it up. Put it back into the stopcock, secure it and start over with the tests. It is better to solve problems in the beginning and not with your product solution in the funnel later on.
Note that it is not necessary to dry the separatory funnel.
4. Performing an
Extraction
a. Place the separatory funnel in an iron ring. Remove the stopper and make sure that the stopcock is closed.
b. Add the solution to be extracted using a short-term funnel.
Do not fill the funnel more than half at this point. Add the
washing/extraction solution and place the stopper on top. There should
still be some room afterwards (75 % of total volume).
c. Take the separatory funnel out of the ring and hold it tightly at the stopper and the stopcock. Invert it slowly and vent (open the stopcock) towards the back of the hood. You will hear a kind of whistle when the pressure is released.
d. Close the stopcock and shake the funnel gently, watching out for emulsions. Vent it again. Repeat this step until no more gas escapes.
e. Place the separatory funnel back in the iron ring. Allow the layers to separate. Then remove the stopper and drain the bottom layer into a clean container. At this point, you need to know which layer contains your desired product.

5. Warnings
If you extract or wash acidic solutions with sodium bicarbonate or sodium carbonate solutions, carbon dioxide will form due to an acid-base reaction. A significant pressure will build up in the funnel. Hence, you need to be more careful in the beginning and vent more often.
H+
+ HCO3-
-----> H2O
+ CO2
If you use low boiling solvents like diethyl ether, pentane, dichloromethane, chloroform, etc. for extraction, you will observe a significant build up of pressure as well.
Due to the pressure build up, you will need to hold on to the stopper and stopcock very tightly. It might be a good idea to wrap a paper towel around the stopper and joint.
When you vent the funnel, point the stem of the funnel away from everybody, so that the solvent and gases released are not blown into somebody's face. The best protocol is to vent it in the back of a hood.
Never throw any layer away, until you are absolutely sure that you isolated your final product. It is easier to isolate it from a small amount of solution than from the waste container.
If you are not sure which layer is organic and which one is aqueous, take a small sample of both layers and add some water, Which layer did increase in volume? Most common organic solvents possess a lower density than water. However, halogenated solvents like dichloromethane, chloroform or carbon tetrachloride are significantly heavier than water (or most diluted aqueous solutions).