
Jones reagent oxidizes a secondary alcohol (such as 2-propanol) to the corresponding ketone (acetone).
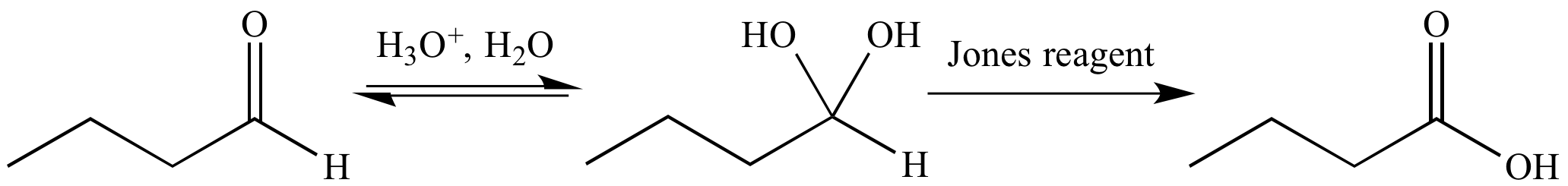
Jones reagent is aqueous and strongly acidic. Under these conditions an aldehyde is in equilibrium with the corresponding geminal diol. Jones reagent oxidizes the geminal diol to a carboxylic acid.
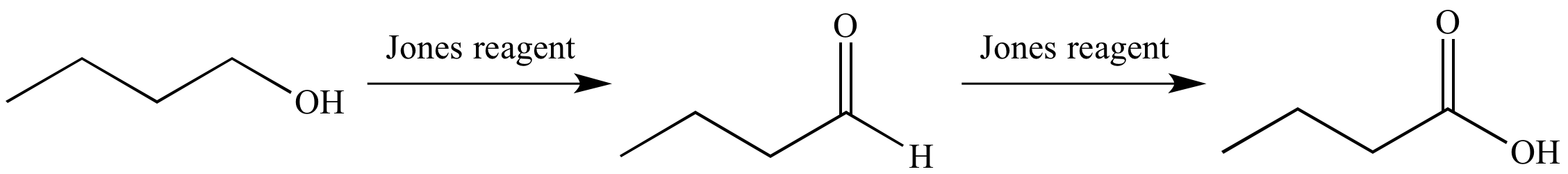
Jones reagent oxidizes a primary alcohol to an aldehyde, and subsequently to a carboxylic acid.