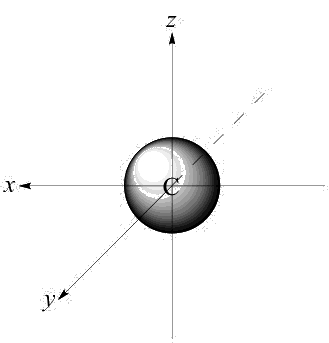
The 2s atomic orbital of carbon is a sphere. It has one node, a hollow spherical 'eggshell' of zero electron density, partway between the nucleus and the outermost edge of the orbital.
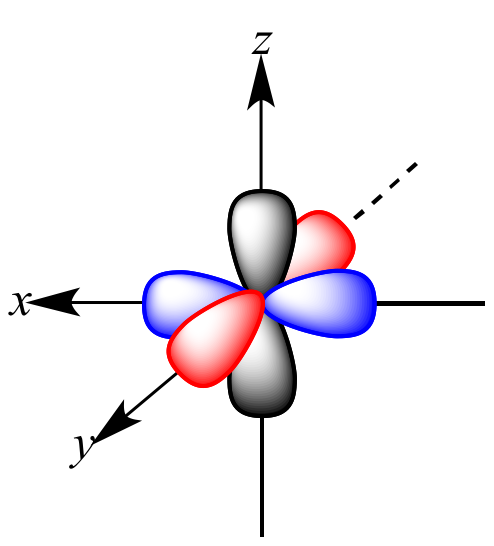
Carbon's 2px, 2py, and 2pz orbitals each have two lobes, and each orbital has one node where the orbital is "pinched off" at the nucleus.