 |
 |
 |
||
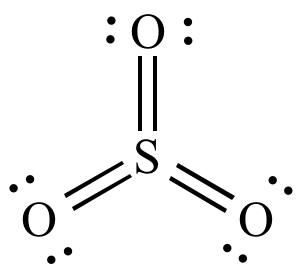
| Lewis
structure |
|
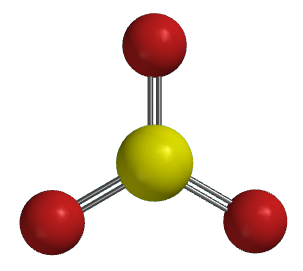
Ball
and spoke |
|
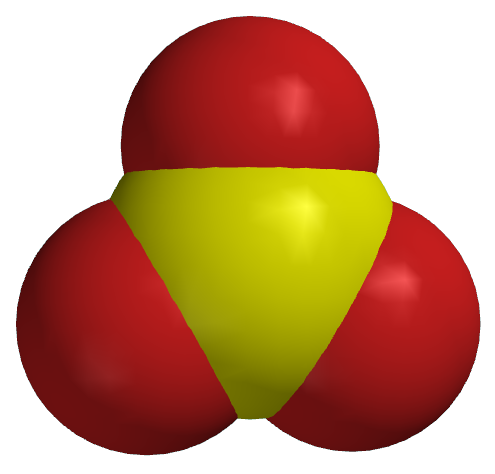
Space
filling
model |
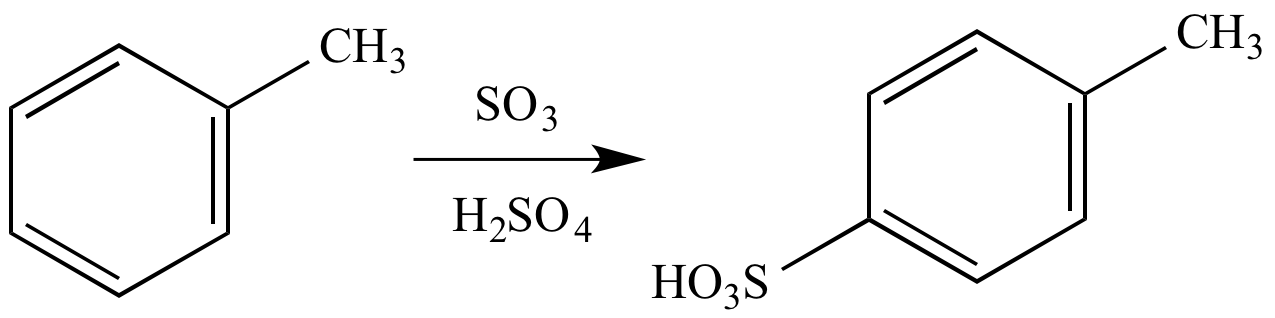
Sulfur trioxide can be used for aromatic sulfonation via the electrophilic aromatic substitution mechanism. In this example, reaction of toluene with sulfur trioxide in sulfuric acid (fuming sulfuric acid or oleum) affords p-toluenesulfonic acid as the major product.