| --video to be added-- |
 |
|
| Click image to see a video
of NH4Cl
dissolving in water. |
|
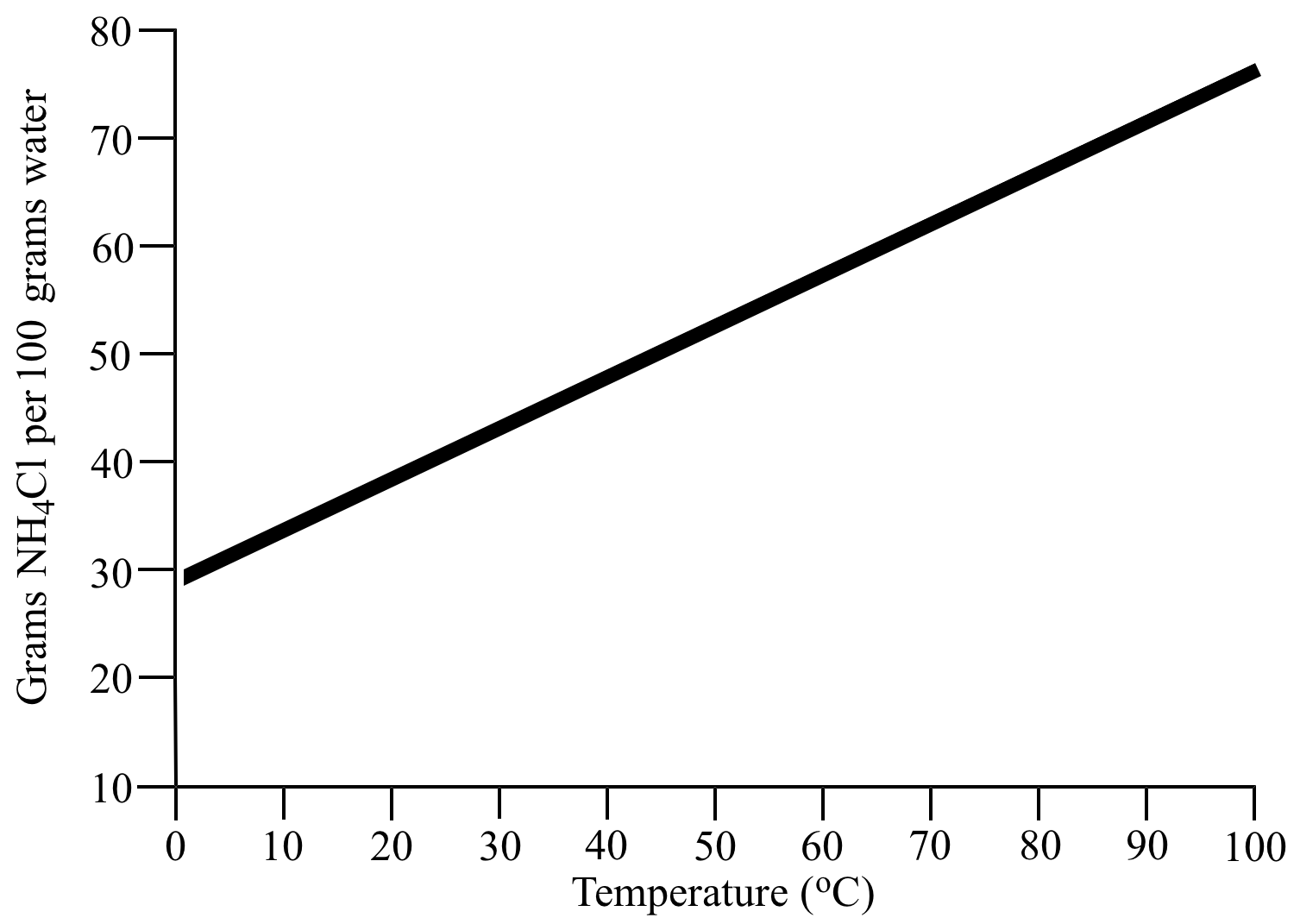
NH4Cl solubility data.
The solubility of NH4Cl
(the solute) in water (the solvent) is 37.2 g NH4Cl per 100 g water at 20 oC. |
 |
<--- Oil layer <--- Vinegar layer (acetic acid + water) |