|
|
Email: mfh@chem.ucla.edu |
The species on the left has 20 boron atoms in it. Those are the mauve spheres. It's a three-minus ion. It forms a very soluble sodium salt. This salt, when dissolved in water, can be placed on the inside of the liposome and then administered to animals, in this case mice bearing EMT-6 adenocarcinoma tumors. You will note the tumor is a mauve square and the tumor line shows the build-up of boron is really quite good. We need something like 20 to 30 parts per million to have a therapeutic dose and here we are peaking out after about 30 hours at 40 parts per million and we're only injecting 10 micrograms of boron per gram of animal. We are actually amplifying the average concentration injected by a factor of four. The nice thing about this is that the blood clears quite readily. You see that the wash-out of blood, liver and spleen are about as you'd expect. This is a good boron agent. We're very excited by this, and it's going into larger animals very shortly.On the right we see a compound that is accommodated in the bilayer. This is an anion, actually a negatively charged group attached to a greasy hydrocarbon tail. It fits nicely in a bilayer membrane. The injected dose is four micrograms of boron per gram of mouse. It's not very much. Now we're again getting above 30 parts per million in tumor even though we're putting in very low injected doses. We observe very good clearance of boron from tissues other than tumor. We're very excited by both of these sets of data. The other thing we're quite excited about is some work that's been pushed by Dr. Bob Kane, in our laboratory, involving what we simply call "trailers" because the proper name is quite cumbersome; oligomeric phosphate diesters. They are actually short chain polymers and they are phosphate esters derived from a phosphate ester linkage and a diol. We synthesize the phosphate diester of a diol and this gives us an oligomer in which we have several of these groups held one to the other n times. We can make these oligomers any size we want by selecting the value of n. 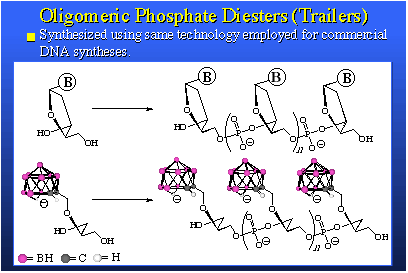
Above here is a stylized view of a DNA molecule. Machines and technology exist to synthesize strings of DNA. Because our starting materials look and behave like DNA, we can use this same technology to make oligomers; trailers. We can put strings of actual DNA onto our oligomers, and vice versa. This is very simple and the versatility that one obtains in the structure of the products is immense. The trailers we synthesize are boron rich, water soluble, bio-compatible, nontoxic, and they present tremendous diversity of structure and secondary chemical function. They can be used in several ways. We can use them as free agents, that is, simply inject them in the animal and let them circulate by themselves, or they can be incorporated within a liposome. Or they can be attached to biomolecules that are themselves targeted for receptor sites in the tumor cell and its DNA. This would involve biomolecule carriers such as hormone surrogates, DNA intercalators, peptides or DNA segments themselves. So, this boron-delivery method has a tremendous scope, both in chemical structure and in use. We're quite excited by our preliminary results. The performance is, so far, astounding. 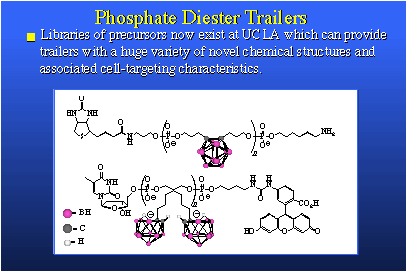
Heretofore, today, we've talked about boron neutron capture therapy using epithermal neutrons. Not too many years ago, fast neutrons were explored as therapeutic radiation and they are still used. They're now used clinically at the University of Washington Medical Center. Fast neutrons having energies, say 25 to 50 million electron volts, are so energetic that they behave like photon beams, which can be shaped. They can be precisely aimed at a target -- a deeply buried tumor in a patient -- and hit that target with great accuracy. Such neutron beams are used therapeutically for certain types of cancer. Prostate cancer is one type. Lung cancer is another. |