|
Recent Journal Covers from the Houk Group Group Member Individual Projects Faculty Research Lecture, 1998 ACS Award in Computers in Chemical and Pharmaceutical Research, 2003 |
Recent Journal Covers and Frontispieces from the Houk Group
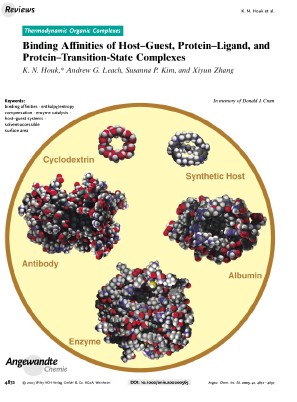
On the frontispiece of Angew. Chem. Int. Ed. 2003, Vol. 42, Issue 40: the CPK models of cyclodextrin, synthetic host, albumin, enzyme and antibody (clockwise). Frontispiece concept and design by Xiyun Zhang and K. N. Houk. In the review paper, we surveyed the affinities of hosts - ranging from small synthetic cavitands to large proteins - for organic molecules. The average association constants for the binding of organic molecules by cyclodextrins, synthetic hosts, and albumins in water, as well as of catalytic antibodies or enzymes for substrates are 103.5±2.5M-1. Binding affinities are elevated to 108±2M-1 for the complexation of transition states and biological antigens by antibodies or inhibitors by enzymes, and to 1016±4M-1 for transition states with enzymes. We explored the origins of such a distribution. In many classes of complexes a rough correlation is found between the binding affinity and the surface area that is buried upon complexation. Enzymes transcend this effect and achieve transition-state binding much greater than is expected from the surface areas.
On the cover of Acc. Chem. Res. 2005, Volume 38, Issue 5: Linus Pauling, Nobel Laureate of both Chemistry and Peace, shown behind an antigen-antibody complex (blue) and an enzyme-transition state complex (red). The bar graphs show the frequencies of occurrence of antibody-antigen complexes (red) and enzyme transition state complexes (blue) versus the binding energies, which increases from left to right. Cover concept and design by K. N. Houk, Xiyun Zhang, and Amy Meyer Phifer. In the paper, we hypothesize that enzymes that accelerate reactions
strongly (by factors of more than 10
Special Edition on Asymmetric Organocatalysis. Guest editors in this number Kendall N. Houk (University of California, Los Angeles) and Benjamin List (Max-Planck-Institut fur Kohlenforschung, Mulheim). On the cover: Four of the prominent organocatalysts described in this issue superimposed over the natural sources or the chiral compounds or precursors. Clockwise from the top left are proline and chicken feather, cinchoinidine and the cinchona flower, Shi's fructose-based catalyst and sugar cane, and a MacMillan organocatalyst and rice hulls (a source of phenylalanine). Cover concept and design by Ruth Gordillo (postdoctoral researcher at Professor Houk's group) and Amy Meyer Phifer (graphic designer). In recent years, catalytic methods based on metal-free organic molecules have experienced a spectacular advancement. Relatively simple organic molecules can be highly effective and remarkably enantioselective catalysts of a wide range of important transformations. Preparative advantages are notable: usually the reactions can be performed under an aerobic atmosphere with wet solvents. The catalysts are inexpensive and they are often more stable than enzymes or other bioorganic catalysts. This issue contains a remarkable number of papers that have been categorized according to a mechanistic classification, where the catalysts are either Lewis acid, Lewis bases, Bronsted acids, or Bronsted bases.
Patrick McCarren and Christopher Suhrada created the design for the cover of the new ACS journal, based on a suggestion by Editor Bill Jorgensen. The pictures and equations included in the montage allude to various concepts in theoretical and computational chemistry, from molecular orbital theory to potential energy surface bifurcations and simulated models of the aqueous environment.
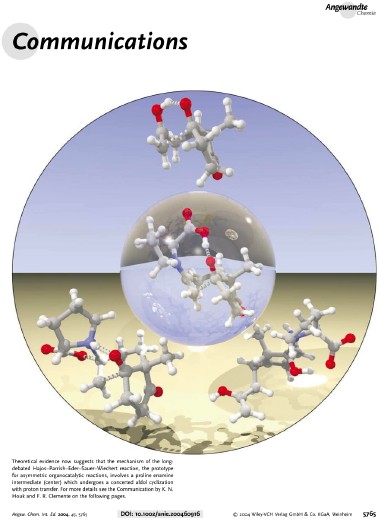
Frontispiece for: Fernando R. Clemene and K. N. Houk, Angew. Chem. Int. Ed. 2004, 43, 5766-5768. Our theoretical studies have clarified the long-debated mechanism of the Hajos-Parrish-Eder-Sauer-Wiechert reaction, the prototype for asymmetric organocatalytic reactions. A comparison of previously proposed models at the C-C bond-forming step with density functional methods indicate that the most favored one involves an enamine intermediate undergoing a concerted aldol cyclization with proton transfer from the proline carboxylic acid group (center). This step is equal in energy to the intramolecular deprotonation leading to the enamine, and both are partially rate-determining steps. |